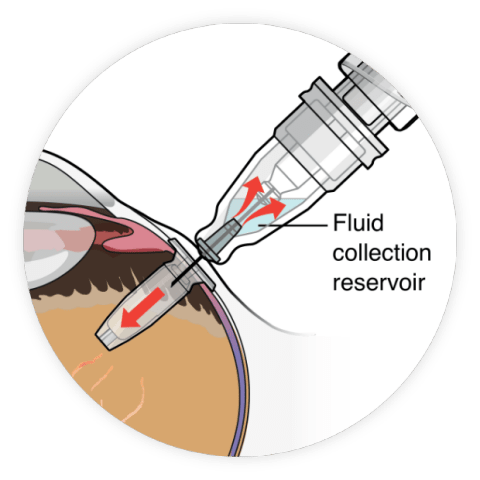
The FDA has approved Roche’s Susvimo (ranibizumab), a long-lasting injectable drug for treating people with “wet” age-related macular degeneration.
Susvimo, which continuously delivers therapy via a refillable ocular implant, can be administered just twice a year, providing an alternative to other injections that are often given monthly.
The drug’s approval was supported by phase 3 data demonstrating that patients treated with Susvimo experienced vision increases equivalent to those receiving monthly ranibizumab injections. Roche noted that 98 percent of patients were able to go six months before needing their first refill of Susvimo.
Additional Information
